Cardiovascular disease remains the leading cause of death globally, despite major advances in prevention and treatment. Elevated low-density lipoprotein cholesterol continues to be one of the most important modifiable risk factors for atherosclerotic cardiovascular disease. Over the past three decades, statins have formed the backbone of lipid management in both primary and secondary prevention. However, a substantial proportion of patients fail to achieve guideline-recommended LDL cholesterol targets, even with high-intensity statin therapy.
This persistent gap in lipid control has driven the development of additional LDL-lowering therapies, including ezetimibe, injectable PCSK9 inhibitors, and more recently, novel oral agents. In February 2026, The New England Journal of Medicine published the CORALreef Lipids trial alongside an editorial examining the clinical significance of enlicitide, a first-in-class oral PCSK9 inhibitor. These publications mark a potentially transformative moment in dyslipidemia management.
This article reviews the scientific rationale, clinical trial evidence, and practical implications of oral PCSK9 inhibition, with a focus on enlicitide and its role in contemporary cardiovascular risk reduction.
LDL cholesterol plays a causal role in the development and progression of atherosclerosis. Large randomized trials and genetic studies consistently demonstrate that lowering LDL cholesterol reduces the risk of myocardial infarction, stroke, and cardiovascular death. Current guidelines therefore emphasize aggressive LDL cholesterol reduction, particularly in patients with established atherosclerotic cardiovascular disease or those at high risk for a first event.
Despite this, real-world data show that up to 70 percent of patients receiving statin therapy do not reach recommended LDL cholesterol goals. Reasons include inadequate statin response, poor medication adherence, intolerance, and therapeutic inertia. As LDL targets have become progressively lower, particularly for secondary prevention, the limitations of statin monotherapy have become increasingly apparent.
Proprotein convertase subtilisin–kexin type 9 regulates LDL receptor degradation in the liver. Inhibition of PCSK9 increases LDL receptor availability and enhances LDL cholesterol clearance from the bloodstream. Injectable monoclonal antibodies targeting PCSK9 have demonstrated LDL reductions of 50 to 60 percent and have been shown to reduce cardiovascular events.
However, real-world adoption of injectable PCSK9 inhibitors has been limited. Barriers include high cost, insurance authorization challenges, the need for subcutaneous injections every two to four weeks, and variable patient acceptance. As a result, only a small fraction of eligible patients currently receive these therapies.
An effective oral PCSK9 inhibitor has the potential to overcome many of these barriers by simplifying treatment, improving adherence, and expanding access to potent LDL-lowering therapy.
Enlicitide is an oral macrocyclic peptide designed to inhibit the interaction between PCSK9 and the LDL receptor. Earlier phase studies demonstrated substantial LDL cholesterol reductions, prompting the large phase 3 CORALreef Lipids trial to evaluate its efficacy and safety over a longer duration and in a broader population.
Unlike small-molecule lipid agents, enlicitide targets the PCSK9 pathway directly, offering LDL-lowering potency comparable to injectable biologics while retaining the convenience of once-daily oral dosing.
The CORALreef Lipids trial was a multinational, randomized, double-blind, placebo-controlled phase 3 study conducted across 168 sites in 14 countries. The trial enrolled adults with either established atherosclerotic cardiovascular disease or elevated risk for a first cardiovascular event.
Eligibility criteria included LDL cholesterol levels of at least 55 mg per deciliter in patients with prior cardiovascular events and at least 70 mg per deciliter in high-risk primary prevention patients. Participants were randomized in a 2 to 1 ratio to receive enlicitide 20 mg daily or placebo for 52 weeks, on a background of standard lipid-lowering therapy.
Importantly, approximately 95 percent of participants were receiving moderate- or high-intensity statins, making the results highly relevant to routine clinical practice.
The primary endpoint was the percent change in LDL cholesterol from baseline to week 24. Secondary endpoints included LDL cholesterol change at week 52 and changes in non-HDL cholesterol, apolipoprotein B, and lipoprotein(a).
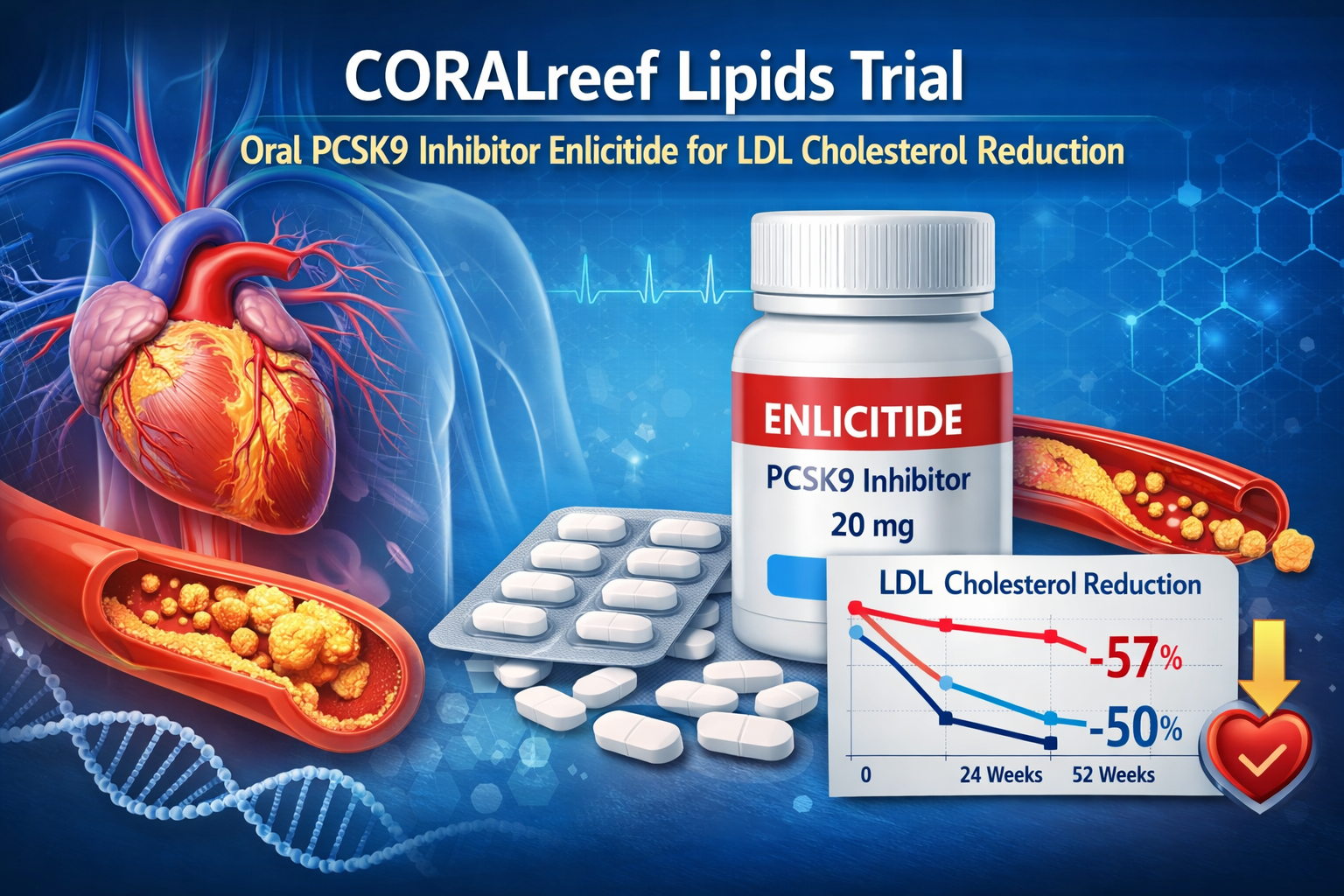
Among more than 2,900 participants, enlicitide produced a mean LDL cholesterol reduction of approximately 57 percent at 24 weeks compared with placebo. At 52 weeks, LDL cholesterol remained nearly 50 percent lower than baseline, demonstrating durability of effect.
Significant reductions were also observed in non-HDL cholesterol and apolipoprotein B, markers that reflect the total burden of atherogenic lipoproteins. Lipoprotein(a) levels were modestly but significantly reduced, consistent with effects seen with other PCSK9-targeting therapies.
Safety outcomes were reassuring. The incidence of adverse events did not differ meaningfully between the enlicitide and placebo groups, and no new safety concerns emerged over one year of follow-up.
Several oral non-statin lipid-lowering agents are currently available, including ezetimibe and bempedoic acid. While these therapies provide incremental LDL reductions, their effects are modest compared with PCSK9 inhibition.
In clinical trials, bempedoic acid has demonstrated LDL cholesterol reductions of approximately 15 to 20 percent, while investigational CETP inhibitors such as obicetrapib have achieved reductions of around 30 percent. In contrast, enlicitide approaches the LDL-lowering efficacy of injectable PCSK9 inhibitors while maintaining oral administration.
This level of potency positions enlicitide as a potential alternative to injectable therapy for many patients who require substantial LDL cholesterol reduction.
If approved, enlicitide could significantly alter lipid management strategies. For patients with established cardiovascular disease who remain above LDL cholesterol targets despite maximally tolerated statins and ezetimibe, an oral PCSK9 inhibitor offers a compelling next step.
High-risk primary prevention patients may also benefit, particularly those with multiple risk factors or genetic dyslipidemia who require aggressive LDL lowering. Improved adherence associated with oral therapy could enhance real-world effectiveness and reduce disparities in cardiovascular outcomes.
However, it is important to note that while LDL cholesterol reduction is a validated surrogate endpoint, definitive evidence of cardiovascular event reduction is still needed.
The ongoing CORALreef Outcomes trial is designed to determine whether enlicitide reduces major adverse cardiovascular events such as myocardial infarction, stroke, and cardiovascular death. The trial is expected to conclude later this decade.
Until these data are available, clinicians must balance the strong biological rationale and LDL-lowering efficacy of enlicitide with the absence of direct outcomes evidence. History suggests that therapies producing large LDL reductions are likely to confer cardiovascular benefit, but confirmation remains essential.
The CORALreef Lipids trial represents a major advance in lipid-lowering therapy. Enlicitide demonstrates that potent PCSK9 inhibition can be achieved through oral administration, potentially addressing long-standing barriers to access and adherence associated with injectable therapies.
While cardiovascular outcomes data are still forthcoming, the magnitude and durability of LDL cholesterol reduction observed with enlicitide position it as a promising addition to the dyslipidemia treatment landscape. If future trials confirm a reduction in cardiovascular events, oral PCSK9 inhibitors may redefine the standard of care for patients at high risk of atherosclerotic cardiovascular disease.
This article is intended for informational and educational purposes only. It does not constitute medical advice, diagnosis, or treatment. Clinical decisions should be based on individual patient circumstances, current clinical guidelines, and consultation with qualified healthcare professionals. The information presented reflects data available at the time of writing and may evolve as new evidence emerges.

Most Accurate Healthcare AI designed for everything from admin workflows to clinical decision support.